
George Khelashvili, Ph.D.
- Associate Professor of Computational Biophysics in the Institute for Computational Biomedicine
1300 York Avenue, Room LC-501A
New York, NY 10065
Techniques
Research Areas
Research Summary:
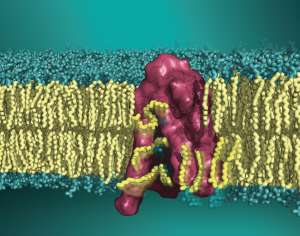
The overall goal of the research projects in the lab is to uncover dynamic mechanisms in fundamental biological processes of signal transduction by cell surface proteins in the categories of receptors (such as G protein-coupled receptors, GPCRs), transporters in the family of Neurotransmitter:Sodium-Symporters (NSS), and lipid scramblases. Special emphasis is on understanding how the spatial organization and function of these molecular machines are regulated by the cell membrane, its components (i.e. cholesterol, various lipids), and interactions with the rich environment of the cell’s proteins. We approach these research topics with advanced quantitative methods of theoretical and computational biophysics, developed and utilized at the highest level of each specialty. We pursue interdisciplinary and multi-scale strategies that integrate biophysical theory and computation with biophysical measurements and molecular cell biology experimentation. Our approach takes advantage of an abundance of molecular level insights from experimental explorations of the function and interactions of membrane-associated signaling proteins, and interprets them in a novel quantitative multi-scale framework to yield insights based on energetics, and experimentally testable hypotheses we validate with respect to mechanisms by which membrane properties and remodeling (e.g. curvature, lipid segregation) affect protein function, organization and signaling-associated interactions that are of major importance to cell physiology.
Recent Publications:
- Feng, Z, Alvarenga, OE, Di Zanni, E, Lee, S, Khelashvili, G, Accardi, A et al.. Calcium dependent activation of the TMEM16F scramblase and ion channel. Nat Struct Mol Biol. 2026;33 (4):664-676. doi: 10.1038/s41594-026-01789-5. PubMed PMID:41998358 PubMed Central PMC13095661.
- Chakraborty, S, Feng, Z, Lee, S, Alvarenga, OE, Panda, A, Zhang, S et al.. Structure and function of the human apoptotic scramblase Xkr4. Nat Commun. 2025;16 (1):7317. doi: 10.1038/s41467-025-62739-1. PubMed PMID:40781244 PubMed Central PMC12334663.
- Kumarage, T, Gupta, S, Morris, NB, Doole, FT, Scott, HL, Stingaciu, LR et al.. Cholesterol modulates membrane elasticity via unified biophysical laws. Nat Commun. 2025;16 (1):7024. doi: 10.1038/s41467-025-62106-0. PubMed PMID:40745155 PubMed Central PMC12313887.
- Marx, DC, Huynh, K, Gonzalez-Hernandez, AJ, Strauss, A, Rico, C, Siepe, D et al.. Structural Diversity and Dynamics of Metabotropic Glutamate Receptor/Beta-Arrestin Coupling. bioRxiv. 2025; :. doi: 10.1101/2025.02.03.636340. PubMed PMID:39975168 PubMed Central PMC11838584.
- Chakraborty, S, Feng, Z, Lee, S, Alvarenga, OE, Panda, A, Bruni, R et al.. Structure and function of the human apoptotic scramblase Xkr4. bioRxiv. 2024; :. doi: 10.1101/2024.08.07.607004. PubMed PMID:39149361 PubMed Central PMC11326236.
- Oh, M, Rosa, M, Xie, H, Khelashvili, G. Automated collective variable discovery for MFSD2A transporter from molecular dynamics simulations. Biophys J. 2024;123 (17):2934-2955. doi: 10.1016/j.bpj.2024.06.024. PubMed PMID:38932456 PubMed Central PMC11393714.
- Doktorova, M, Khelashvili, G, Ashkar, R, Brown, MF. Molecular simulations and NMR reveal how lipid fluctuations affect membrane mechanics. Biophys J. 2024;123 (9):1164. doi: 10.1016/j.bpj.2024.04.018. PubMed PMID:38679022 PubMed Central PMC11079997.
- Carten, JD, Khelashvili, G, Bidon, MK, Straus, MR, Tang, T, Jaimes, JA et al.. A Mechanistic Understanding of the Modes of Ca2+ Ion Binding to the SARS-CoV-1 Fusion Peptide and Their Role in the Dynamics of Host Membrane Penetration. ACS Infect Dis. 2024;10 (2):398-411. doi: 10.1021/acsinfecdis.3c00260. PubMed PMID:38270149 .
- Menon, I, Sych, T, Son, Y, Morizumi, T, Lee, J, Ernst, OP et al.. A cholesterol switch controls phospholipid scrambling by G protein-coupled receptors. J Biol Chem. 2024;300 (2):105649. doi: 10.1016/j.jbc.2024.105649. PubMed PMID:38237683 PubMed Central PMC10874734.
- Menon, I, Sych, T, Son, Y, Morizumi, T, Lee, J, Ernst, OP et al.. A cholesterol switch controls phospholipid scrambling by G protein-coupled receptors. bioRxiv. 2024; :. doi: 10.1101/2023.11.24.568580. PubMed PMID:38045315 PubMed Central PMC10690279.
- Bergman, S, Cater, RJ, Plante, A, Mancia, F, Khelashvili, G. Substrate binding-induced conformational transitions in the omega-3 fatty acid transporter MFSD2A. Nat Commun. 2023;14 (1):3391. doi: 10.1038/s41467-023-39088-y. PubMed PMID:37296098 PubMed Central PMC10250862.
- Doktorova, M, Khelashvili, G, Brown, MF. Efficient calculation of orientation-dependent lipid dynamics from membrane simulations. bioRxiv. 2024; :. doi: 10.1101/2023.05.23.542012. PubMed PMID:37292992 PubMed Central PMC10245828.
- He, C, Mansilla-Soto, J, Khanra, N, Hamieh, M, Bustos, V, Paquette, AJ et al.. CD19 CAR antigen engagement mechanisms and affinity tuning. Sci Immunol. 2023;8 (81):eadf1426. doi: 10.1126/sciimmunol.adf1426. PubMed PMID:36867678 PubMed Central PMC10228544.
- Mlynarczyk, C, Teater, M, Pae, J, Chin, CR, Wang, L, Arulraj, T et al.. BTG1 mutation yields supercompetitive B cells primed for malignant transformation. Science. 2023;379 (6629):eabj7412. doi: 10.1126/science.abj7412. PubMed PMID:36656933 PubMed Central PMC10515739.
- Doktorova, M, Khelashvili, G, Ashkar, R, Brown, MF. Molecular simulations and NMR reveal how lipid fluctuations affect membrane mechanics. Biophys J. 2023;122 (6):984-1002. doi: 10.1016/j.bpj.2022.12.007. PubMed PMID:36474442 PubMed Central PMC10111610.
- Khelashvili, G, Kots, E, Cheng, X, Levine, MV, Weinstein, H. The allosteric mechanism leading to an open-groove lipid conductive state of the TMEM16F scramblase. Commun Biol. 2022;5 (1):990. doi: 10.1038/s42003-022-03930-8. PubMed PMID:36123525 PubMed Central PMC9484709.
- Cheng, X, Khelashvili, G, Weinstein, H. The permeation of potassium ions through the lipid scrambling path of the membrane protein nhTMEM16. Front Mol Biosci. 2022;9 :903972. doi: 10.3389/fmolb.2022.903972. PubMed PMID:35942471 PubMed Central PMC9356224.
- Morra, G, Razavi, AM, Menon, AK, Khelashvili, G. Cholesterol occupies the lipid translocation pathway to block phospholipid scrambling by a G protein-coupled receptor. Structure. 2022;30 (8):1208-1217.e2. doi: 10.1016/j.str.2022.05.010. PubMed PMID:35660161 PubMed Central PMC9356978.
- Zhang, X, Xie, H, Iaea, D, Khelashvili, G, Weinstein, H, Maxfield, FR et al.. Phosphatidylinositol phosphates modulate interactions between the StarD4 sterol trafficking protein and lipid membranes. J Biol Chem. 2022;298 (7):102058. doi: 10.1016/j.jbc.2022.102058. PubMed PMID:35605664 PubMed Central PMC9207681.
- Kots, E, Mlynarczyk, C, Melnick, A, Khelashvili, G. Conformational transitions in BTG1 antiproliferative protein and their modulation by disease mutants. Biophys J. 2022;121 (19):3753-3764. doi: 10.1016/j.bpj.2022.04.023. PubMed PMID:35459639 PubMed Central PMC9617077.

